
1.0 Introduction
An 11 months old male infant, David Black was admitted to the hospital due to an episode of bloody diarrhoea (hematochezia), after which he appeared to be very pale. He had a previous history of severe reflux and fussy feeding behavior in response to breast milk and it was eased by the reduction of dairy products from his mother’s milk. Eczema was noted along with small petechiae from a few weeks of age on patient’s arms, legs and became infected with Staphylococcus aureus. As well, he has been treated for upper respiratory tract infections at the ages of 5, 7,& 9 months. Further conversation with patient’s parents revealed, patient had contracted more respiratory infections and they had thought this unremarkable as his older siblings attended day care.
On examination the patient was afebrile, alert, active but distressed and tugging his ear. He had a mild pale pulse rate of 92 bpm, was found to be small for his age, eczematous rash over the scalp and limbs. His ears were visualized and left ear found to be inflamed. Laboratory investigations were carried out in order to diagnose the possible underlying cause.
1.1 Peripheral blood film (PBF)
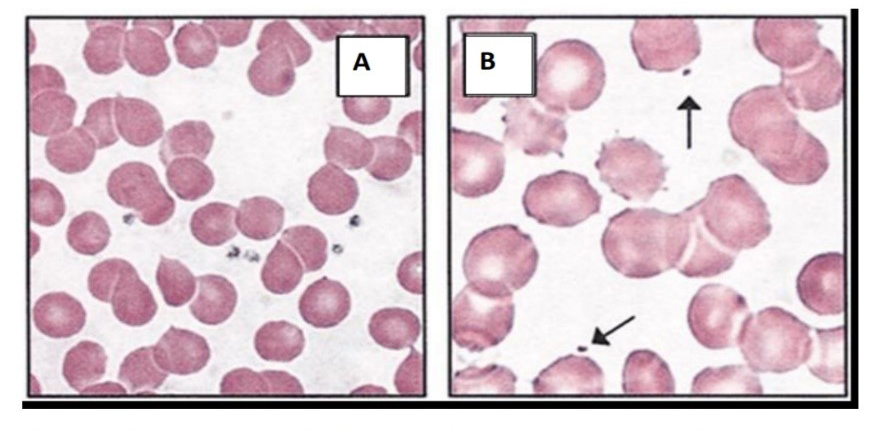
Figure 1: Peripheral blood film of the patient (A shows normal control, B shows the patient’s sample and arrow indicate platelets).
1.2 Full blood count (FBC)
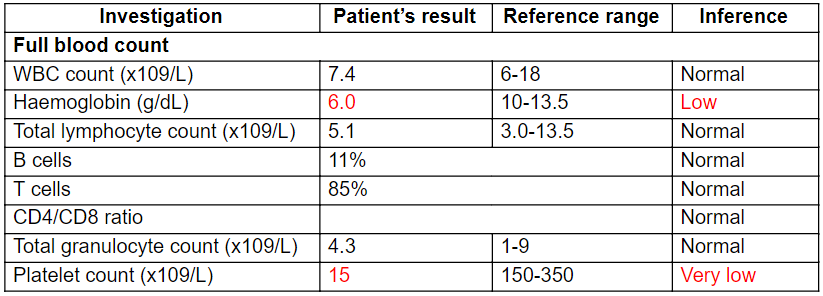
Table 1: Patient’s results obtained from FBC
1.3 Immunoglobulin test
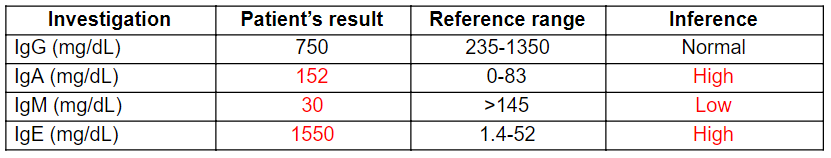
Table 2: Patient’s results obtained from Immunoglobulin test
1.4 Additional test results
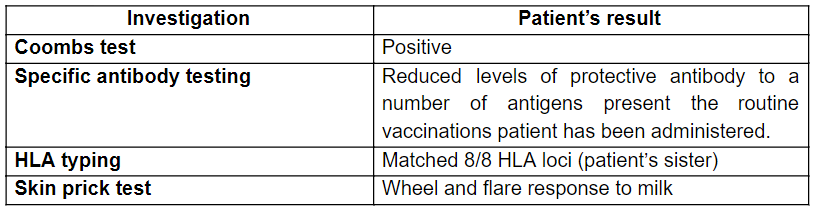
Table 3: Additional investigations
2.0 Diagnosis and discussion
2.1 Clinical suspicions/differential diagnosis related to the clinical presentation by the patient
The patient was admitted to the hospital due to an episode of bloody diarrhea after which he appeared to be very pale. Hematochezia in infants mainly suggests a serious disease, most likely suggestive of milk protein allergy (MPA), intestinal infection/gastroenteritis which is caused by enteric pathogens, Wiskott-Aldrich-Syndrome (WAS), hyper IgE syndrome (HIES) (Paediatrics, 2014; Murphy, 2008). The pallor after the episode of hematochezia may be due to a possible anaemic condition due to blood loss or dehydration due to fluid loss as a consequence of diarrhoea (Brueton et al., 2007).
Considering the patient’s clinical symptoms, an FBC and a PBF was carried out to assess the patient’s general health and possible anaemic condition. FBC is usually a quantification of cellular components in the blood in order to check for the abnormalities in haematological parameters (Nabil, 2018). Usually leukocyte count increases in an infection but the patient’s FBC revealed a normal leukocyte count excluding the possibility of gastroenteritis due to an enteric infection (Medlineplus, 2018). Patient’s hemoglobin level showed a reduction supporting possible anaemic condition and is more suggestive of anaemia due to blood loss than the fluid loss due to diarrhoea. Hemoglobin level usually reduces in iron deficiency anaemia/hemolytic anaemia (Barrell, 2017). A PBF analysis is usually carried out for the cytological examination of patient’s blood smear in order to check whether there are any abnormalities in blood cellular components (Adewoyin and Nwogoh, 2014). The patient’s PBF revealed hemolysis further suggesting the possibility of hemolytic anaemia.
The patient had a previous history of severe reflux and fussy feeding behavior in response to breast milk and it was eased by the reduction of dairy products from his mother’s milk. Fussiness and severe regurgitation during breast feeding is indicative of hypersensitive reaction against breast milk further suggesting the possibility of MPA. Since the medical history revealed that the infant’s fussy feeding behavior was eased via the management of diary content in mother’s diet, MPA could be the possible underlying cause for the patient’s medical history (Brueton et al., 2007). Moreover, WAS also results in food allergies due to aberrations in T-reg regulation (Gudjonsson and Liang, 2016)
Patient’s medical history also revealed that during few weeks of age, the patient had eczema in his limbs along with small petechiae and became infected with Staphylococcus aureus causing infected eczema. He had also previously been treated for URTIs at 5, 7 and 9 months indicating recurrent infections. Eczema, secondary bacterial infections with Staphylococcus aureus and recurrent infections are more common in patients with MPA, WAS, Common variable immunodeficiency (CVID), hyper IgE syndrome (HIES) and X-linked agammaglobulinaemia (XLA) (Schwartz, 2018).
In MPA, in response to milk proteins the body generates a hypersensitive reaction giving rise to eczematous rash in infants. The patient’s WAS are more prone to develop eczema as a secondary complication (Morita, 2018; Schwartz, 2018; Freeman, Heimall and Holland, 2010; Brueton et al., 2007).
Infected eczema may occur with the entering of Staphylococcus aureus which is found in normal skin flora. It can enter via open blisters/wounds at the site of eczematous rash causing a secondary bacterial infection (Fletcher, 2017). Based on the symptoms, for further analysis immunoglobulin test and skin prick test was done.
Immunoglobulin test is a quantitative test that is done to assess the level of immunoglobulins (Lab tests online, 2017). Patient’s results revealed a normal IgG, elevated IgA, low IgM and highly elevated IgE, excluding the possibility of XLA since IgG is usually undetectable in XLA (Schwartz, 2018). Whereas, in HIES, IgE level should be >or= 2000. But the patient’s IgE level was 1550 excluding the possibility of HIES (Abraham et al., 2009). In CVID IgA, IgG and IgM levels reduces and therefore CVID can also be excluded (Schwartz, 2018). The findings of this test mainly supportive of WAS and IgE-mediated MPA (Schwartz, 2018; Holm et al., 1999). A SPT is done to measure the IgE level in response to suspected food allergen (Caffarelli et al., 2013). The patient’s SPT showed wheel and flare response to milk protein confirming MPA. Specific antibody testing revealed the reduction in protective antibodies such as IgM and other antibodies are low for certain number of specific antigens.
The endothelial damages to blood vessels are usually regulated by hemostasis which is mediated by thrombocytes. The formation of petechiae is mainly suggestive of possible internal bleeding due to haemorrhage. Hence, it is indicative of severe thrombocytopenia (Natchman and Raffi, 2008). The patient’s FBC revealed a severe reduction in platelets while the PBF also showed a reduction in platelet number confirming thrombocytopenia. As well, PBF revealed a reduction in platelet size further supporting WAS in which micro-thrombocytopenia is a prominent feature (Aiuti et al., 2009).
On admission the patient was afebrile, alert, active but distressed and tugging his ear. He had a mild pale pulse rate of 92 bpm and was diagnosed with failure to thrive (FTT). Alertness indicates the sleeping issues in patient while his distressed condition may be manifested by tachypnea as a result of hypoxia due to the reduced hemoglobin level in patient (Hermansen and Lorah, 2007). Mild pale pulse rate of the patient may also be due to reduced Hemogobin. Reduced growth in the patient may be due to the elimination of all the dairy products from the mother’s/infant’s diet without appropriate substitutions which can lead to malnutrition and diarrohea may cause FTT in patient due to MPA and FTT occurs in WAS as well (Lifschitz and Szajewska, 2015; Cole and Lanham, 2011; Belleau et al., 2004). Moreover, FTT may occur as a result of anaemia (Bines and Israel, 1991).
Further examination revealed eczematous rash over the scalp, limbs and the left ear was found to be inflamed further supporting WAS. Previously patient had eczema in his limbs and now an eczematous rash has progressively developed and spread to the scalp indicating chronic anaemia and suggestive of blood loss beyond hematochezia since PBF was also revealed hemolysis of red cells. Left ear was inflamed upon examination and this may be due to the spreading of eczema into the ear. Moreover, ear infections are more common in WAS (Schwartz, 2018).
A coombs test and was done for the further confirmation of anaemic condition of the patient. Direct coombs test is based on the RBC agglutination by anti-human antibodies and usually done to investigate autoimmune hemolytic anaemia (AHA) (Karp, 2016). The patient’s result indicated coombs positive confirming AHA.
Inclusion or exclusion of clinical suspicions were done with the aid of test results (Table 1). The results obtained from the investigations indicated possible autoimmune disease (Wiskott-Aldrich-Syndrome) correlated with milk protein allergy as the underlying cause for patient’s signs and symptoms. WAS is characterized by immunodeficiency, micro-thrombocytopenia and eczema. Moreover it leads to autoimmune manifestations such as autoimmune hemolytic anaemia and recurrent infections. Hence, patient’s condition is complicated with WAS and MPA.
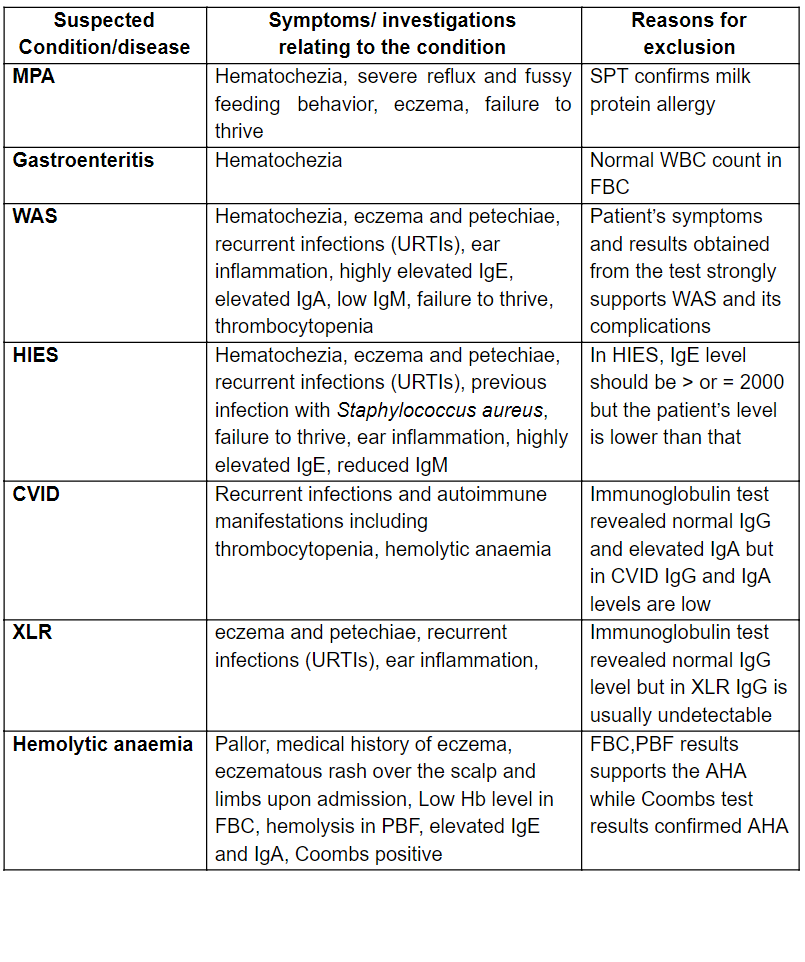
Table 4: Exclusion/inclusion of differential diagnosis
2.2. General immune mechanism for infectious disease
WAS is a severe X-linked primary immunodeficiency which is caused by mutations in gene encoding WAS protein (WASP). WASP is a main regulator in cytoskeletal reorganization in hematopoietic cells. WASP deficiency causes multiple defects in all immune cells leading to immunodeficiency, micro-thrombocytopenia and eczema (Figure 2). Moreover it leads to food allergies, recurrent infections and autoimmune manifestations such as hemolytic anaemia (Bosticardo et al., 2012).

Figure 2: Schematic view of cellular defects described in WAS-deficient immune cells (Bosticardo et al., 2012)

Figure 3: Schematic view of immunodeficiency and autoimmunity in WAS (Bosticardo et al., 2012).
In WASP-deficiency, both the innate and adaptive immunity impairs leading to immunodeficient and autoimmune complications. Functional defects in macrophages, DCs, T-cell, B-cells lead to incomplete clearance of pathogens and making WAS patients more susceptible for infections. Reduced CD21 and CD35 which are complement receptors that involves in antigen presentation and negative selection of self-reactive B-cells. Expression of WASP-deficient T-regs leads to loss of tolerance in T-cell and B-cell regulation. Defects in process of restimulation-induced cell death leads to defective T-cell death after the re-stimulation of TCR synchronizing persistent T-cell response pathogens/self-antigens. Defective B-cells results in autoimmunity via production of auto-antibodies in WAS (Bosticardo et al., 2012).
The mechanism of micro-thrombocytopenia is not fully understood. It could be due to increased exposure of phosphatidylserine which are present on cell membranes of WASP-deficient platelets leading to aberrant clearance of thrombocytes or autoimmune elimination of thrombocytes by antiplatelet antibodies. Immunopathogenesis of eczema in WAS is not clearly understood. However, hyper-IgE levels and aberrant functioning of T-cells in WASP-deficient patients may play a causative role in eczema. Mechanism of autoimmune hemolytic anaemia in WAS is also not fully clarified and it could be due to incomplete pathogen clearance and bystander tissue damage which occur as a result of chronic inflammation. As well, it could be due to loss of tolerance to self-antigens and abberant functioning of T-reg cells (Morita, 2018; Bosticardo et al., 2012; Aiuti et al., 2009).
Dysregulation of Th2 occurs due to the lack of control over T-reg cells and cause food allergies in WAS patients initiating allergic sensitization to dietary antigens. Moreover, in WAS IgE-mediated reactions occur against common food allergens causing inflammation (Gudjonsson and Liang, 2016) (Figure 4). Inflammatory DCs process the allergen and present it to CD4+ T-lymphocytes via MHC-II. In exposure to IL-4 CD4+ T-cells differentiate into pro-allergic Th2 cells initiating the activation of B-lymphocytes. Contemporaneously, there is an impairment in the regulation of T-reg cells and it exerts no suppressive effects on Th2 cell activity leading to continuous stimulation of B-lymphocytes. It is believed that deregulation of T-reg cells and lack of polarization of allergen specific T-cells towards the Th2 cells precede the B-cells drive. Subsequently, the interaction between B-cells with IL-4 and IL-13 initiates the immunoglobulin class switching eventually producing IgE antibodies (Garssen et al., 2014).
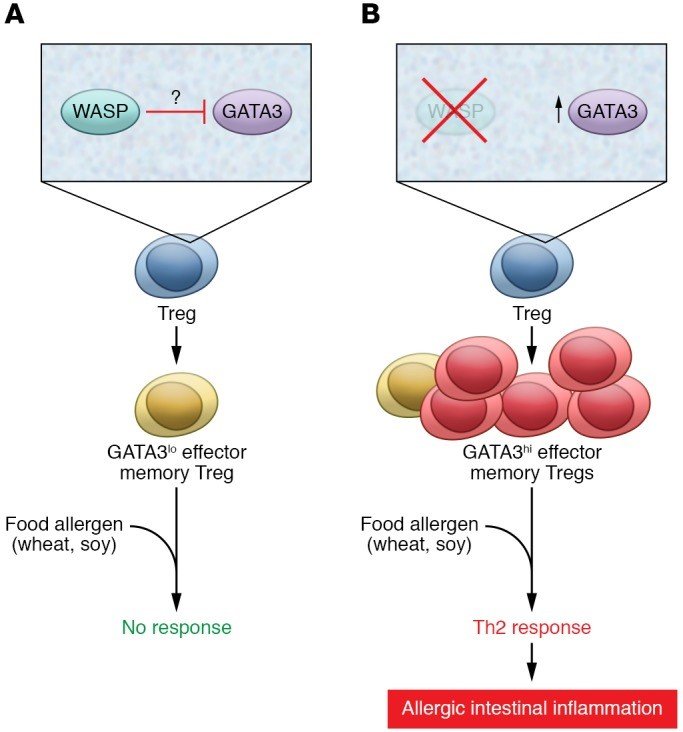
Figure 4: Schematic diagram of food allergy in WAS
FA shows the In WASP expressing T-regs in which the GATA3 expression is suppressed and the thereby prevent Th2 mediated inflammation in exposure to dietary antigens. B shows the WASP-deficient T-reg which is unable to repress the expression of GATA3 and thereby mediate the Th2 allergic inflammation in response to dietary antigens (Gudjonsson and Liang, 2016).
2.3. Further investigations
Based on the patient’s clinical presentation and the results obtained from the laboratory investigations it was evident that the patient is suffering from Wiskott-Aldrich-Syndrome correlated with milk protein allergy. However, further investigation needed to be carried out for a definite confirmation of diagnosis (Table 4).
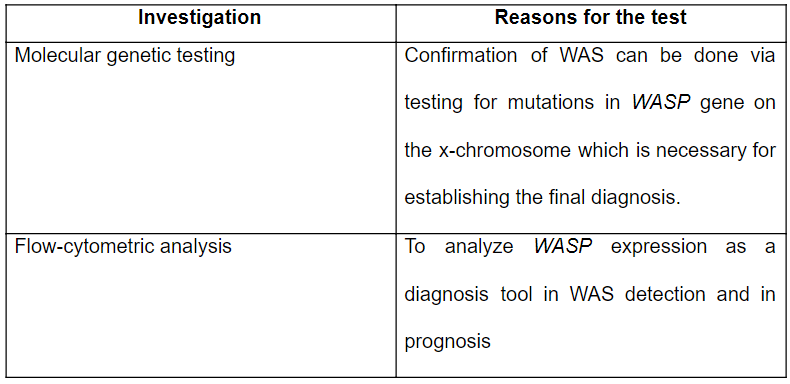
Table 5: Further confirmatory tests that are needed to be carried out (Porter, 2017; Buchibinder, Fillipovich and Nugent, 2014)
3.0 Treatment plan
Based on the patient’s diagnosis several treatment plans are suggested shown below in table in order to regain his quality of life. HLA typing of the patient matched 8/8 HLA loci with one of his sisters. Therefore it is possible to perform a bone marrow transplant from the sister to the patient in order to treat WAS since it is the available curative in both genetic and immunological complications of WAS (Kirchhausen and Rosen, 1996). Anti-rejection drugs and anti-microbial drugs are also prescribed to prevent complications.

Table 6: Suggestive treatment plan (Hyunh, 2017; Kaveri and Patel, 2013; Notarangelo and Pai, 2011)
4.0 Conclusion
Patient’s symptoms and medical history led to many clinical suspicions. With the support of the test results obtained, the patient’s symptoms were supported in order to rule in or rule out the differential diagnosis. The patient’s symptoms were linked with many complications and the results indicated possible Wiskott-Aldrich-Syndrome correlated with milk protein allergy as the underlying cause for the patient’s symptoms. Patient management and precise treatments are recommended to rehabilitate a healthy life.
5.0 Abbreviations
AHA Autoimmune hemolytic anemia
BMT Bone marrow transplant
CVID Common variable immunodeficiency
FTT Failure to thrive
HIES Hyper-IgE syndrome
IVIG Intravenous gammglobulin
MPA Milk protein allergy
PBF Peripheral blood analysis
SPT Skin prick test
WAS Wiskott-Aldrich-Syndrome
XLR X-linked gammaglobulinaemia
6.0. References
Abraham, R.S., Boyce, T.G., Hagan, J.B., Iyer, V.N., Joshi, A.Y. and Park, M.A. (2009) ‘Elevated serum immunoglobulin E (IgE): when to suspect hyper IgE syndrome- A-10-year pediatric tertiary care center experience’, Allergy and Asthma Proceedings, 30(1), pp.23-27.
Adewoyin, A.S. and Nwogoh, B. (2014) ‘Peripheral blood film: A review’, Annals of Ibadan Postgraduate Medicine, 12(2), pp.71-79.
Aiuti, A., Bosticardo, M., Marangoni, F., Roncarolo, M.G. and Villa, A. (2009) ‘Recent advances in understanding the pathophysiology of Wiskott-Aldrich-Syndrome’, Blood, 113, pp.6288-6295.
Alfina, C., Carla, C., Domenico, A., Elena, L. and Giovanna, V. (2012) ‘The immunopathogenesis of cow’s milk protein allergy (CMPA)’, Italian Journal of Pediatrics, 38(35).
Barrell, A. (2017) Medscape. Available in: https://www.medicalnewstoday.com/articles/318050.php (Accessed: 21 July 2018).
Belleau, J.T., Conley, M.E., Hernandez-Trujillo, V.P., Jeng, M., Lew, D.B. and Nguyen, W.T. (2004) ‘Cow’s milk protein allergy in a patient with hyper-IgE syndrome’, Annals of Allergy, Asthma and Immunology, 92(4), pp.469-474.
Bines, J.E. and Israel, E.J. (1991) ‘Hypo-proteinemia, anemia and failure to thrive in an infant’, Gastroenterology, 101(3), pp. 848-856.
Bishara, J., Bivas, A., Gafter-Givili, A., Leibovici, L., Mansur, N., Paul, M., Zemer-Wessercug, N. (2011) ‘Thrombocytopenia in Staphylococcus aureus bacteremia: Risk factors and prognostic importance’, Mayo Clinic Proceedings, 86(5), pp.389-396.
Bosticardo, M., Castiello, A.C., Catucci, M., Pala, F. and Villa, A. (2012) ‘Autoimmunity in Wiskott-Aldrich-Syndrome: an unsolved engima’, Frontiers in Immunology, 3(1), pp.1-14.
Bruenton, M., Dupont, C., Hill, D., Isolauri, E., Koletzko, S., Oranje, A.P., Staiano, A. and Vandenplas, Y. (2007) ‘Guidelines for the diagnosis and management of cow’s milk protein allergy in infants’, Archives of Disease in Childhood, 92, pp.902-908.
Buchibinder, D., Fillipovich, A.H. and Nugent, D.J. (2014) ‘Wiskott-Aldrich syndrome: diagnosis, current, management and emerging treatments’, The Application of Clinical Genetics, 7, pp.55-66.
Buda, A. and Miedzobrodzki, J. (2016) ‘The role of Staphylococcus aureus in secondary infections in patients with atopic dermatitis’, Polish Journal of Microbiology, 65(3), pp.253-259.
Cole, S.Z. and Lanham, J.S. (2011) ‘Failure to thrive: An update’, American Family Physician, 83(7), pp.829-834.
Davis, C.P. (2017) MedicineNet. Available at: https://www.medicinenet.com/is_eczema_contagious/article.htm (Accessed: 14 July 2018).
Deshpande, P.G. (2017) Medscape. Available at: https://emedicine.medscape.com/article/974019 (Accessed: 14 July 2018).
Fletcher, J. (2017) Medical News Today. Available at: https://medicalnewstoday.com/articles/319561 (Accessed: 13 July 2018).
Freeman, A., Heimall, J. and Holland, S.M. (2010) ‘Pathogenesis of hyper IgE syndrome’, Clinical Review of Allergy and Immunology, 38(1), pp.32-38.
Garssen, J., Jo, J., Knipples, L. and Sandalova, E. (2014) ‘Role of cellular immunity in cow’s milk allergy; Pathogenesis, tolerance induction and beyond ’, Mediators of Inflammation, 2014, pp.1-10.
Hermansen, C.L. and Lorah, K.N. (2007) ‘Respiratory distress in the new born’, American Family Physician, 76(7), pp.987-994.
Holm, K., Kautiainen, H., Majamaa, H., Moisio, P. and Turjanmaa, K. (1999) ‘Cow’s milk allergy: diagnostic accuracy of skin prick test and patch tests and specific IgE’, Allergy, 54(4), pp.346-351.
Hyunh, P.N. (2017) Medscape. Available at: https://emedicine.medscape.com/article/137015#d5 (Accessed: 01 March 2018).
Jaakkola, J.J.K., Jaakkola, M.S. and Rantala, A. (2013) ‘Respiratory infections in adults with atopic disease and IgE antibodies to common aeroallergens, PLOS one, 8(7), pp. 1-7.
Karp, J.K. (2016) Medscape. Available at: https://emedicine.medscape.com/article/1731264-overview#a1 (Accessed: 21 July 2018).
Kaveri, S.V. and Patel, V. (2013) ‘The mechanisms of action of IVIG in autoimmune and inflammatory diseases’, International Society of Blood Transfusion, 8, pp.185-188.
Kirchhausen, T. and Rosen, F.S. (1996) ‘Disease mechanism: Unravelling Wiskott-Aldrich-Syndrome’, Current Biology, 6(6), pp.676-678.
Lab tests online (2017) AACC. Available at: https://labtestsonline.org/tests/quantitative-immunoglobulins (Accessed: 21 July 2018).
Lai, F. and Yang, Y. (2017) ‘The prevalence and characteristics of cow’s milk protein allergy in infants and young children with iron deficiency anemia’, Pediatrics and neonatology, Elsevier [Online]. DOI: https://doi.org/10.1016/j.pedneo.2017.01.004 (Accessed: 14 July 2018).
Lifschitz, C. and Szajewska, H. (2015) ‘Cow’s milk allergy: evidence based diagnosis and management for the practitioner’, European Journal of Pediatrics, 174, pp.141-150.
Mokha, J. and Serrano, M. (2012) ‘Thrombocytopenia associated with cow’s milk protein allergy’, Clinical Pediatrics, 52(10), pp.985-987.
Morita, A. (2018) Medscape. Available at: https://emedicine.medscape.coom/article/1114511-overview#a4 (Accessed: 21 July 2018).
Murphy, S.M. (2008) ‘Management of bloody diarrhea in children in primary care’, British Medical Journal, 336(7651), pp.1010-1015.
Nabil, S.N. (2018) Medicine Net. Available at: https://www.medicinenet.com/complete_blood_count/article.htm# (Accessed: 19 July 2018).
Natchman, R. L. and Raffi, S. (2008) ‘Platelets, petechiae and preservation of the vascular wall’, New England Journal of Medicine, 359(12), pp. 1261-1270.
National Eczema Society (2017) Atopic eczema. Available at: http://www.eczema.org/atopic-eczema (Accessed: 17 July 2018).
Notarangelo, L.D. and Pai, S. (2011) ‘Hematopoietic cell transplantation for Wiskott-Aldrich-Syndrome: Advances in biology and future directions for treatment’, Immunology and allergy clinics of North America, 30(2), pp.179-194.
Paediatrics (2014) Childhood Diarrhoea. Available at: https://patient.info/doctor/childhood-diarrhoea (Accessed: 13 July 2018).
Schwartz, R. A. (2018) Medscape. Available at: https://emedicine.medscape.com/article/1050956-clinical (Accessed: 21 July 2018).
Schwartz, R.A. (2018) Medscape. Available at: https://emedicine.medscape.com/article/1051103-overview#a1 (Accessed: 31 July 2018).
Tiercy, J. (2016) ‘How to select the best available or unrelated donor of hematopoietic stem cells’, Haematologica, 101(6), pp.680-687.




